Groundbreaking Treatment Boosts Survival Rates for Hodgkin Lymphoma
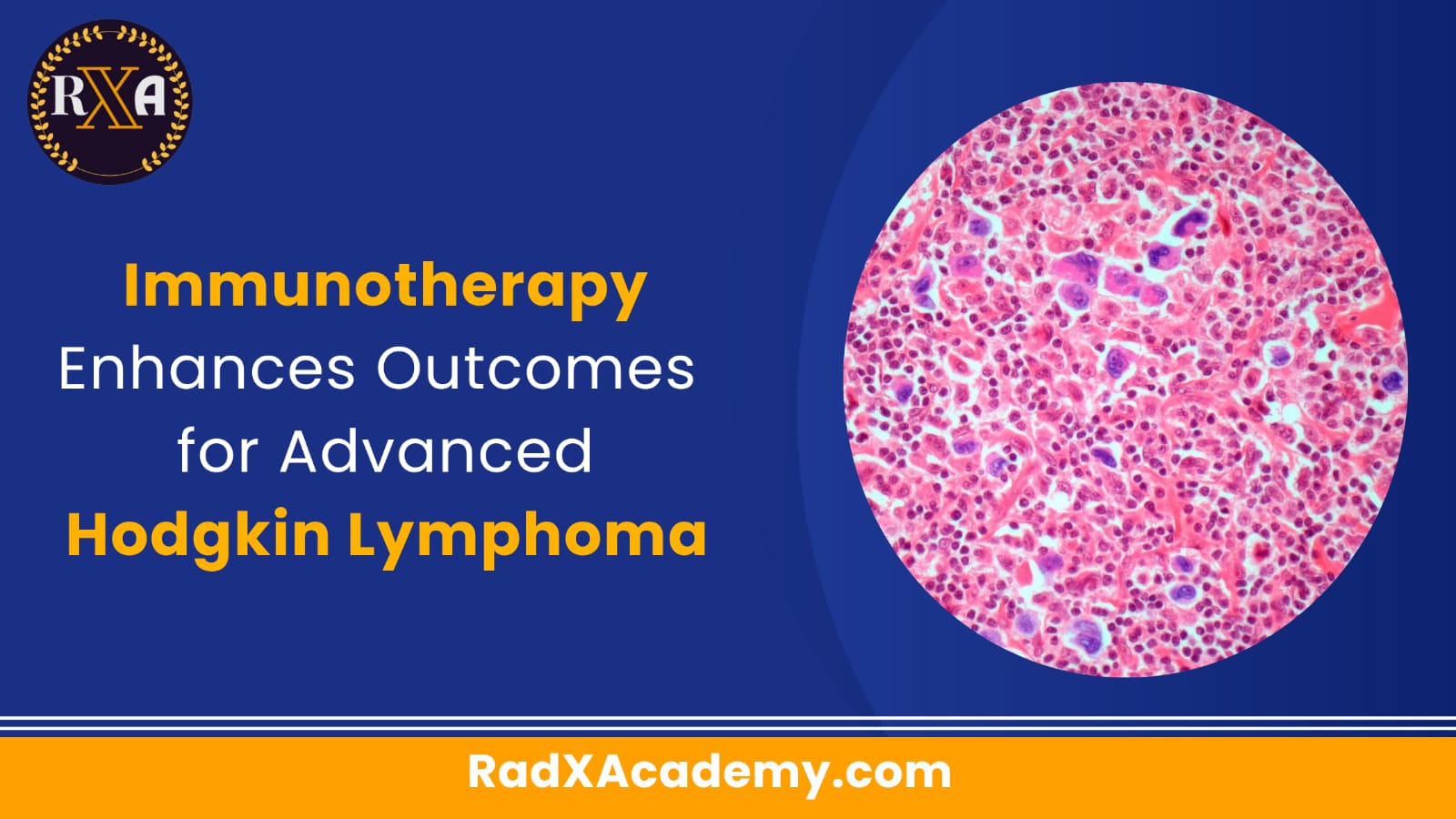
An innovative cancer treatment that supercharges the immune system has significantly improved survival rates for advanced Hodgkin lymphoma, raising the two-year survival rate to a remarkable 92%. Published in the prestigious New England Journal of Medicine, the study heralds a potential new standard of care for the disease.
Hodgkin Lymphoma and Its Impact
Hodgkin lymphoma, a rare form of blood and immune system cancer, disproportionately affects young people and is classified under lymphomas. Despite the high cure rates associated with existing therapies, long-term side effects, including secondary cancers, infertility, and damage to the heart and lungs, remain serious concerns.
“We will see fewer breast cancers decades later, less infertility, and less heart disease in this patient group,” said Dr. Jonathan Friedberg, director of the Wilmot Cancer Institute at the University of Rochester Medical Center and lead investigator of the study.
Traditional Treatment vs. New Approaches
Current therapies for Hodgkin lymphoma, which include chemotherapy and often radiation, cure over 80% of patients. However, for the remaining 20%, the outlook is more challenging, with a higher risk of treatment-related complications.
The goal of this groundbreaking clinical trial, led by Dr. Friedberg, was to not only improve survival rates but also reduce long-term toxicities—an achievement that has earned praise across the oncology community.
Innovative Immunotherapy Breakthrough
The phase 3 clinical trial, identified as S1826, enrolled nearly 1,000 patients aged 12 and older through the SWOG Cancer Research Network. Participants were divided into two groups: one receiving chemotherapy alongside brentuximab vedotin, the current standard treatment, and the other receiving chemotherapy with the immunotherapy drug nivolumab, which specifically targets a common genetic mutation in Hodgkin lymphoma.
After two years of follow-up, the trial results showcased:
- 92% survival without disease progression in the immunotherapy group, compared to 83% in the standard care group.
- Reduced side effects among patients treated with nivolumab, as the study avoided radiation therapy—a method historically associated with long-term complications like secondary cancers and organ damage.
A Collaborative Effort
This large-scale study exemplifies the power of cooperative research, involving contributions from dozens of researchers across the U.S. and Canada. Notable collaborators include Dr. Alex Herrera of City of Hope, biostatisticians Dr. Michael LeBlanc and Hongli Li from the Fred Hutch Cancer Center, and pediatric oncologists Dr. Kara Kelly and Dr. Sharon Castellino from leading cancer research institutions.
Dr. Friedberg emphasized the inclusivity of the trial, which enrolled:
- Pediatric patients as young as 12,
- Older adults up to 60+, and
- 25% participants from underrepresented groups.
Such inclusivity has made the study a model for future trials, particularly those targeting adolescents and young adults with cancer.
Expedited Path to Standard Care
The preliminary data was so compelling that the National Cancer Institute (NCI) halted the trial early to expedite the review process by the U.S. Food and Drug Administration (FDA). Already approved for other indications, nivolumab is expected to be rapidly integrated into treatment guidelines for stage 3 and 4 Hodgkin lymphoma.
Dr. Friedberg and his team’s landmark findings promise to reshape how Hodgkin lymphoma is treated, offering hope to thousands of patients worldwide and setting a new benchmark in cancer care.